Explain Why Three Different Solvent Mixtures Were Used Column Chromatography
The seven mixtures are as follows. There are different ways to separate mixtures for example by filtration crystallisation distillation or chromatography.
If the Rf 05 you probably should use a smaller amount of ethyl acetate and re-run the TLC.
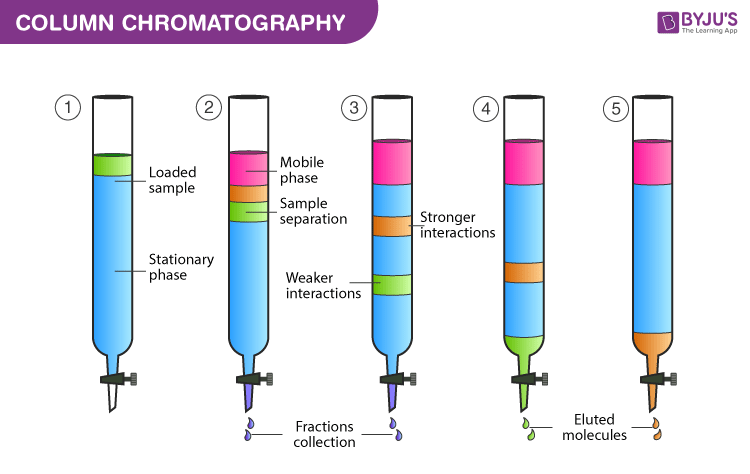
. Gel column chromatography In this method of chromatography the separation takes place through a column packed with gel. Column chromatography is used by an organic chemist to separate liquids and solids from a solution. You run a column to separate a mixture of three compounds.
In thin layer chromatography the stationary phase is a thin layer of silica gel or alumina on a glass metal or plastic plate. 1 isocratic and 2 gradient. We leverage these differences to distribute molecules between a stationary phase and a mobile phase.
For example 70-230 silica gel is used for gravity columns and 230-400 mesh for flash columns. Up to 256 cash back Why was itimportant to mark the TLC plate with pencil. Because of these differences some components of the mixture stay longer in the stationary phase and they move slowly in the chromatography system while others pass rapidly into mobile phase and leave the system faster 3.
The solute selected should have the ability to dissolve the components of the mixture. The principle behind column chromatography is adsorption in which a mixture of. Column chromatography CC is one of the most useful methods for the separation and purification of both solids and liquids.
Which wavelengths of light were mostabsorbed in your leaves. Here you mix both liquid and solid outside of the column and pour it into the column. 2 5 ml hexane.
Note the pigments present in the ink are all water soluble. There are two types of solvent systems are used in column chromatography. Chromatography relies on two different phases.
You may need touse both the TLC and spectrophotometricdata. Column chromatography will take longer because of the larger quantities of chemicals that are used. It helps in the separation of impurities from any kind of mixture.
At the point when the mobile phase together with the mixture that requires to be isolated is brought in from the top of the column the movement of the individual. The column is run with a solvent system based on hexanes but which becomes more polar with the addition of more and more dichloromethane over time. The main principle involved in column chromatography is the adsorption of the solutes of the solution with the help of a stationary phase and afterward separates the mixture into independent components.
Using informationfrom the prelab lecture determine which pigments are present inyour various leaf samples. 5 ml ethyl acetate. The method chosen depends upon the type of mixture.
Sometimes these columns are several inches. Adsorbent particle size affects how the solvent flows through the column. The mobile phase is the solvent that moves through the paper carrying different substances with it.
Naphthalene o-toluic acid and fluorenol. This mixture flows down the column and the component of the sample is separated by splitting between the mobile eluent and stationary. In the first portion of the experiment TLC was used to separate compounds.
In thin layer chromatography the stationary phase is a thin layer of silica gel or alumina on a glass metal or plastic plate. The process can be automated as well. Varieties of solvent can be used in the process of separation based on the required results.
Column chromatography is more effective at separating compounds because it can be. Sometimes more than one solvent will be used to elute a mixture from a column beginning with a nonpolar solvent to elute the nonpolar compounds and then changing to a polar solvent to elute the polar compounds. Instead of spreading a thin layer of the stationary phase on a plate the solid is packed into a long glass column either as a powder or a slurry.
Smaller particles higher mesh values are used for flash chromatography larger particles lower mesh values are used for gravity chromatography. Based on this approach three components form the basis of the chromatography technique. There are two common ways you can fill a chromatography column.
In column chromatography the silica can be used dry or in a slurry. Various sizes of chromatography columns are used and if you follow a link at the bottom of the. Solvents are used to help separate components of a mixture.
The same stationary phase and the same mobile phase can be used. For this method add your dry solid phase to the column and pass equilibria buffer or starting solvent to saturate the solid. The water may be acidified or basified to optimize the method.
Published August 29 2016. Any variety and quantity separation are possible through column chromatography. Column chromatography works on a much larger scale by packing the same materials into a vertical glass column.
Here is a video of an experiment conducted to separate the components of water soluble ink. A column of absorbents which is silica gel or it may be alumina gets loaded in a column filled with impurities when experimenting. This is a solidliquid technique in which the stationary phase is a solid and the mobile phase is a liquid.
Column chromatography allows you to separate larger quantities of material than TLC. What color were your leaves to startwith. Common mobile phases are a mix of acetonitrile and water or water and methanol.
Partition column chromatography The stationary phase as well as mobile phase are liquid in partition chromatography. In liquid chromatography the mobile phase is generally a mix of polar and non-polar or less polar solvents. Various sizes of chromatography columns are used and if you follow a link at the bottom of the.
Seven different solvent mixtures were compared in order to evaluate which mixture was most effective as an elution solvent in separating four unknown compounds. It works just like TLC. Run a TLC plate in 82 pet etherEtOAc ethyl acetate Measure the Rf of the desired compound.
How Separation Works in Column Chromatography Methods. The stationary phase is a solvent held in the gap of a solvent. The reason for use of a sequential solvent process in chromatography is that it increases the efficiency of the separation.
In an isocratic system only one solvent is passed through the stationary phase but in the gradient system. It helps in the separation of any mixture. Molecules vary in size charge polarity and solubility.
Column chromatography is another kind of liquid chromatography. Column chromatography works on a much larger scale by packing the same materials into a vertical glass column. Two solvents with predefined ratios have been passed through the stationary phase to separate the components.
But because molecules are so different its not possible. Sep 22 2014. Predict the elution order.
Separation in column chromatography relies on differences.

Chem Is Fun Lab Separation Of A Mixture By Paper Chromatography Paper Chromatography Matter Science Chemistry Lessons

Column Chromatography Principle Procedure Applications Elution In Chromatography

Principles Of Chromatography Stationary Phase Article Khan Academy

Column Chromatography Types Principle Instrumentation And Application Chemistry Notes

Comments
Post a Comment